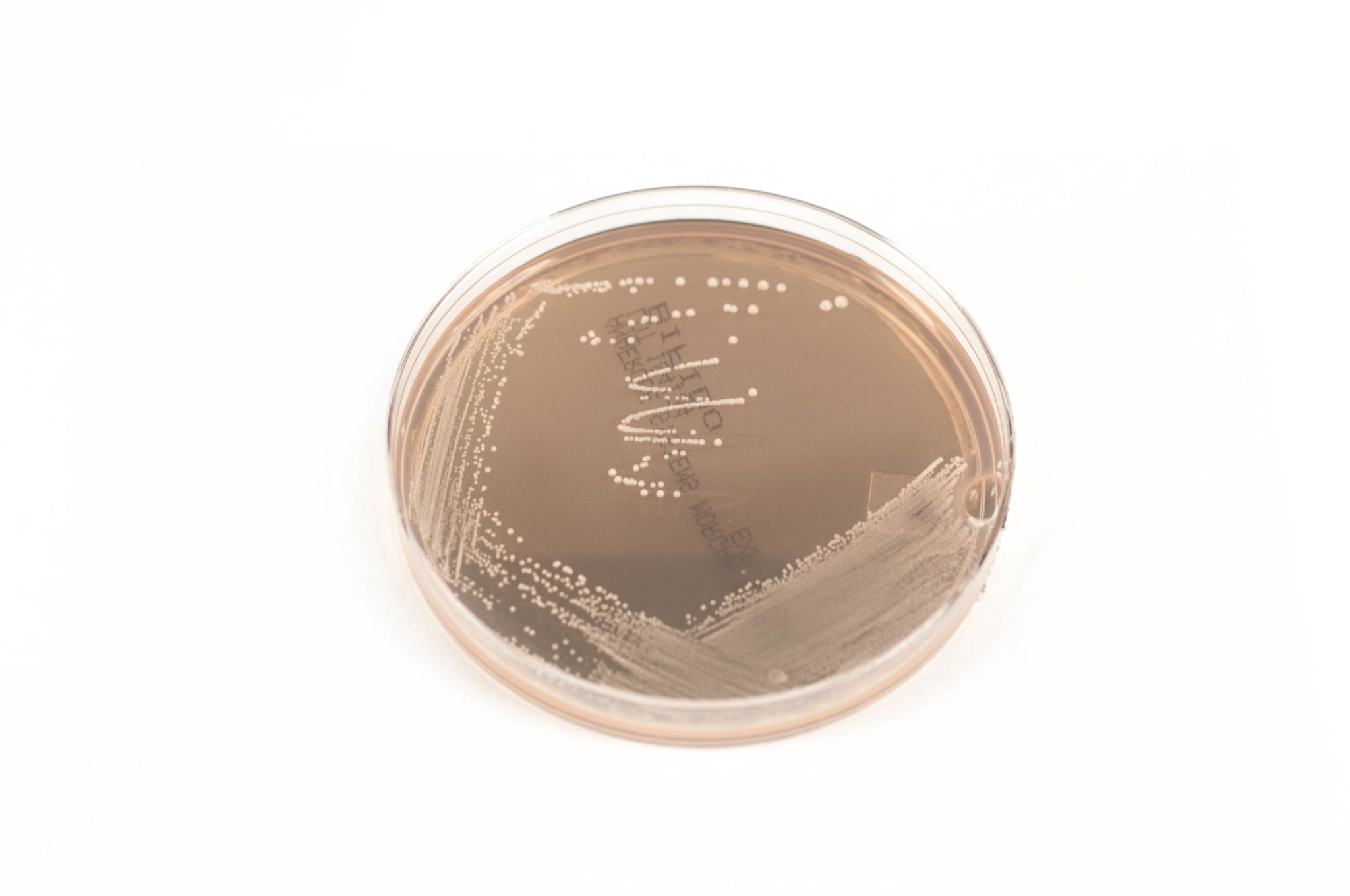
Bifidobacterium Selective Agar (BIFIDO) is a selective media for the isolation and enumeration of Bifidobacterium species.
Bifidobacterium Selective Agar – BIFIDO
| Catalog # |
Description |
Qty | Price |
| AS-6423 | Bifidobacterium Selective Agar (BIFIDO) | 4 plates | $13.16 |
Non-stock item: 25 pack minimum order, allow 2-4 weeks for delivery.
Product Description & Summary
BIFIDO agar contains a reinforced clostridial agar formulation as the basal nutritive media. Selective and differential compounds include iodoacetic acid and 2, 3, 5-triphenyltetrazolium chloride (TTC). Iodoacetate inhibits glyceraldehyde-3-phosphate dehydrogenase, drastically reducing the growth of non-bifidobacterial colonies. TTC makes it possible to differentiate between other species and Bifidobacterium, since the bifidobacterial develop in large white colonies. This media is prepared, dispensed, and packaged under oxygen-free conditions to prevent the formation of oxidized products prior to use.
Interpretation of Results & Limitations
This media supports good growth of some of the Bifidobacterium spp. found in clinical infections.
BIFIDO will not provide complete information for identification of bacterial isolates. Additional test procedures and media are required for complete identification. It is recommended that a non-selective media, such as Brucella Blood Agar (BRU, catalog #: AS-111) also be inoculated from the same clinical specimen to assure recovery of all species present. Consult reference materials for additional information.
Quality Control
The following organisms are routinely used for quality control testing at Anaerobe Systems.
| Organism Tested | ATCC # | Results | Time |
| Bacteroides fragilis | 25285 | Growth | 24 – 48 hrs |
| Fusobacterium nucleatum | 25586 | No Growth | |
| Clostridium perfringens | 13124 | Inhibited | |
| Peptostreptococcus anaerobius | 27337 | Variable | |
| Proteus mirabilis | 12453 | Inhibited | |
| Staphylococcus aureus | 25923 | Variable | |
| Escherichia coli | 25922 | No Growth | |
| Bifidobacterium longum | 15707 | Growth | 48 – 72 hrs |
| Bifidobacterium bifidum | 15696 | Growth | 48 – 72 hrs |
| Bifidobacterium adolescentis | 15703 | Growth | 48 – 72 hrs |
| Bifidobacterium breve | 15700 | Variable | 48 – 72 hrs |
Product Storage & Shelf Life
Storage: Upon receipt, store at room temperature in original package until used. Avoid overheating or freezing. Do not use media if there are signs of deterioration (shrinking, cracking, or discoloration due to oxidation of media) or contamination. The expiration date applies to the product in its original packaging and stored as directed. Do not use product past the expiration date shown on the label.
Shelf Life: 90 days from date of manufacture.